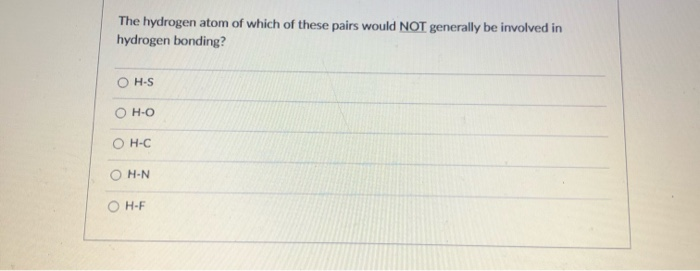
(For example, H2O has 2x1 + 6 8 valence electrons, CCl4 has 4 + 4x7. This work simulates the real working state of the fuel cells and provides significant guidance for rapid start-up in anion exchange membrane fuel cells (AEMFCs). Ionic bonds result from a transfer of electrons from one species (usually a metal). The resultant AEM shows outstanding fuel cell power density (210 mW cm −2) under special operating conditions like 40 ☌ and 50% RH, which exhibits 2.5 times better than hydrogen bonding-free AEM. This means that carbon dioxide is less soluble in water than polar. Besides, the hydrogen bond network formed by urea-urea group and urea-water group endows the AEMs with excellent water retention ability, thereby decreasing the dependence of OH − conduction on RH. CO2 can form hydrogen bonds with water, but its linear shape makes it a nonpolar molecule. Microstructure analysis and molecular dynamics simulation show that hydrogen bonding between urea-based structures enhances the self-assembly ability of quaternary ammoniums groups and expanded OH − conduction pathway. Herein, we report an AEM with urea-based structure that exhibits excellent low humidity performance. Molecules with F-H, O-H, or N-H moieties are very strongly attracted to similar moieties in nearby molecules, a particularly strong type of dipole-dipole.  
The apparent degradation of alkaline fuel cells performance at reduced relative humidity (RH) drives the interest in further improving anion exchange membranes (AEMs) functionalities and/or developing alternative AEMs.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
